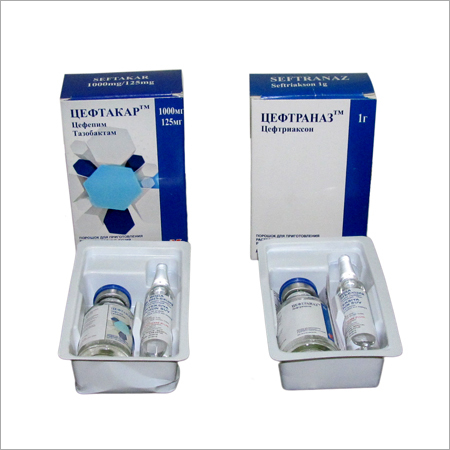
Seftakar & Seftranaz
Product Details:
- Dosage Form Injection
- Salt Composition Ceftriaxone 1000 mg + Tazobactam 125 mg
- Origin of Medicine India
- Pacakaging (Quantity Per Box) 1 Vial + 1 Ampoule of Sterile Water for Injection
- Indication Treatment of susceptible bacterial infections
- Drug Type Prescription
- Ingredients Ceftriaxone and Tazobactam
- Click to View more
X
Seftakar & Seftranaz Price And Quantity
Seftakar & Seftranaz Product Specifications
- To be administered by a registered medical practitioner
- Ceftriaxone and Tazobactam
- As directed by physician
- Injection
- Adults and Children
- Ceftriaxone 1000 mg + Tazobactam 125 mg
- India
- Store in a cool and dry place below 25C, protect from light
- 1 vial per box
- Antibiotic
- Bacterial Infections
- Prescription
- Powder for Injection
- 1 Vial + 1 Ampoule of Sterile Water for Injection
- Treatment of susceptible bacterial infections
- To be reconstituted with provided sterile water before use
- Rx Only (Prescription Required)
- 24 months from manufacturing date
- Cephalosporin Antibiotic with Beta-lactamase Inhibitor
- Seftakar & Seftranaz
- Known hypersensitivity to cephalosporins or beta-lactam antibiotics
- Intravenous or Intramuscular as specified
Seftakar & Seftranaz Trade Information
- Telegraphic Transfer (T/T), Cheque
- Depends On requirement Week
- Yes
- Contact us for information regarding our sample policy
- Customized Packaging
- Australia, North America, Eastern Europe, Middle East, Western Europe, Africa, Central America, South America, Asia
Product Description
| ANTIBIOTIC/ ANTIFUNGAL / ANTIMALARIAL / ANTI - INFECTIVES | |||
| Azithromycin 250mg | TABLET | ||
| Azithromycin 500mg | TABLET | ||
| Ofloxacin 200mg | TABLET | ||
| Ofloxacin 400mg | TABLET | ||
| Ofloxacin 200mg + Ornidazole 500mg | TABLET | ||
| Cefpodoxime 50mg | TABLET | ||
| Cefpodoxime 100mg (Dispersible Tablet) | TABLET | ||
| Cefpodoxime 200mg | TABLET | ||
| Cefpodoxime 200mg + Clavulanic Acid 125mg | TABLET | ||
| Cefpodoxime 200mg + Azithromycin 250mg | TABLET | ||
| Cefpodoxime 200mg + Dicloxacillin 500mg (ER) | TABLET | ||
| Cefpodoxime 200mg + Levofloxacin 250mg | TABLET | ||
| Cefixime Trihydrate 50mg | TABLET | ||
| Cefixime Trihydrate 100mg (Dispersible Tablet) | TABLET | ||
| Cefixime Trihydrate 200mg | TABLET | ||
| Cefixime 50mg + Clavulanic Acid 31.25mg | TABLET | ||
| Cefixime 100mg + Clavulanic Acid 62.5mg | TABLET | ||
| Cefixime 200mg + Clavulanic Acid 125mg | TABLET | ||
| Cefixime Trihydrate 200mg + Dicloxacillin 500mg (Ext. Rel.) | TABLET | ||
| Cefixime 200mg + Ofloxacin 200mg | TABLET | ||
| Cefixime 200mg + Ornidazole 500mg | TABLET | ||
| Cefixime 200mg + Azithromycin 250mg | TABLET | ||
| Amoxycillin 200mg + Clavulanic Acid 28.5mg | TABLET | ||
| Amoxycillin 250mg + Clavulanic Acid 125mg | TABLET | ||
| Amoxycillin 500mg + Clavulanic Acid 125mg | TABLET | ||
| Amoxycillin 875mg + Clavulanic Acid 125mg | TABLET | ||
| Cefuroxime 250mg | TABLET | ||
| Cefuroxime 500mg | TABLET | ||
| Cefuroxime 250mg + Clavulanic Acid 125mg | TABLET | ||
| Cefuroxime 500mg + Clavulanic Acid 125mg | TABLET | ||
| Linezolid 600mg | TABLET | ||
| Levofloxacin 250mg | TABLET | ||
| Levofloxacin 500mg | TABLET | ||
| Levofloxacin 750mg | TABLET | ||
| Ciprofloxacin 500 mg | TABLET | ||
| Amoxycillin 250mg + Dicloxacillin 250mg | CAPSULE | ||
| Cefpodoxime 50mg / 5ml | DRY SYRUP | ||
| Cefpodoxime 50mg + Clavulanic Acid 31.25mg/5ml | DRY SYRUP | ||
| Cefixime 50mg/5ml | DRY SYRUP | ||
| Cefixime 50mg + Clavulanic Acid 31.25mg/5ml | DRY SYRUP | ||
| Amoxycillin 200mg + Clavulanic Acid 28.5mg/5ml | DRY SYRUP | ||
| Cefpodoxime 100mg/5ml | DRY SYRUP | ||
| Azithromycin Suspension 100mg | SUSPENSION | ||
|
| |||
Broad-Spectrum Antibacterial Protection
Seftakar & Seftranaz utilize a robust combination of Ceftriaxone and Tazobactam, targeting a wide range of bacterial pathogens. This combination enhances antibiotic effectiveness against both Gram-positive and Gram-negative bacteria by preventing bacterial resistance, making it a reliable choice for complex infections.
Convenient Powder for Injection Formulation
Each vial is packaged with a matching ampoule of sterile water, designed for on-the-spot reconstitution. This ensures high potency and stability while allowing administration via intravenous or intramuscular injection, facilitated by medical professionals for precision and safety during therapy.
Stringent Storage and Shelf Life Guidelines
Seftakar & Seftranaz are formulated to maintain stability for up to 24 months when stored correctly below 25C and protected from light. Careful adherence to storage requirements preserves the drugs effectiveness, offering peace of mind to healthcare providers and patients alike.
FAQs of Seftakar & Seftranaz:
Q: How should Seftakar & Seftranaz be administered?
A: Seftakar & Seftranaz are intended for intravenous or intramuscular injection. The powder must be reconstituted with the supplied sterile water by a registered medical practitioner before administration to ensure proper dosage and effectiveness.Q: What is the recommended storage condition for this medication?
A: The vials should be stored in a cool, dry place below 25C and protected from direct light. Adhering to these conditions helps maintain the medications stability and potency throughout its 24-month shelf life.Q: Who can use Seftakar & Seftranaz, and for what kind of infections are they prescribed?
A: These antibiotics are suitable for both adults and children and are indicated for the treatment of bacterial infections susceptible to Ceftriaxone and Tazobactam, as determined by a physician.Q: When should the product be reconstituted, and what should be used for this process?
A: The injection should be reconstituted immediately before use, using only the sterile water ampoule provided in the packaging. This ensures the medication remains stable and effective until administration.Q: What are the primary ingredients in Seftakar & Seftranaz, and how do they work?
A: Each vial contains Ceftriaxone (1000 mg) and Tazobactam (125 mg). Ceftriaxone kills bacteria by disrupting their cell wall synthesis, while Tazobactam inhibits beta-lactamase enzymes that some bacteria use to resist antibiotics.Q: Are there any contraindications for using this product?
A: Seftakar & Seftranaz should not be used in patients with known hypersensitivity to cephalosporins, beta-lactam antibiotics, or any ingredients in the formulation. Inform your doctor of any allergies before starting treatment.Q: Can the medicine be used without a prescription, and who should administer it?
A: No, Seftakar & Seftranaz are prescription-only medications (Rx Only) and must be used under medical supervision. Administration should be performed by a registered medical practitioner to ensure correct reconstitution and dosage.Tell us about your requirement

Price:
Quantity
Select Unit
- 50
- 100
- 200
- 250
- 500
- 1000+
Additional detail
Mobile number
Email
Other Products in 'ANTIBIOTIC/ ANTIFUNGAL / ANTIMALARIAL' category
 |
KARAN HEALTHCARE PVT. LTD.
All Rights Reserved.(Terms of Use) Developed and Managed by Infocom Network Private Limited. |